- PET/SPECT Products
- - Tropane analogs
- - PET/SPECT Products
- Metal Chelating Agent
- - Bifunctional Chelating Agents
- - Functionalized Macrocycles
- - CheMatech Products
- Peptides
- - RGD Peptides
- - ApoPep-1
- - DOTA-TATE, TOC, NOC
- - TAT Peptides
- Chemicals
- - Paclitaxel
- - Chemicals
- - Resins
- Disposable Ware
- - Radio-Cap
- PSMA
- Imidazolium-based ionic liquids
- - 1,1,2,2,-tetrafluoroethanesulfonate
- - 1,1,2,3,-tetrafluoroethanesulfonate
- - 1,1,2,4,-tetrafluoroethanesulfonate
- - 1,1,2,5,-tetrafluoroethanesulfonate
- - 1,1,2,6,-tetrafluoroethanesulfonate
- - 1,1,2,7,-tetrafluoroethanesulfonate
- - 1,1,2,8,-tetrafluoroethanesulfonate
- - Acetate
- - Bis(trifluoromethylsulfonyl)imides
- - Bromides
- - Chlorides
- - Dicyanamides
- - Hexafluorophosphates
- - Iodides
- - Methylcarbonates
- - Nitrate
- - Phosphates
- - Sulfonates, Sulfates
- - Tetrachloroferrates
- - Tetrahiocyanatocobaltate
- - Thiocyanates
- - Tricyanomethanides
- - Trifluoroacetate
- - Trifluoromethanesulfonate
- - tetrafluoroborate
- Quarternary Ammonium Salts
- Piperidinium-based ionic liquids
- Pyridinium-based ionic liquids
- - Bis(trifluoromethylsulfonyl)imides
- - Bromides
- - Chlorides
- - Hexafluorophosphates
- - Iodies
- - Perfluorobutanesulfonate
- - Tetrafluoroborates
- - Triflates
- Pyrrolidinium-based ionic liquids
- Ammonium-Based Ionic Liquids
- Phosphonium-based ionic liquids
- Sulfonium-based ionic liquids
- DABCO-Based ionic liquids
- Speciality for Ionic Liquids
- Ionic liquids Screening Kits
- N-Protecting Reagents
- Peptide Coupling Reagents
- Linkers for Solid Phase Synthesis
- Other Reagents
- Amino Acids and Derivatives
- Unusual Amino Acids
- N-Methyl Amino Acids
- Boc-Amino Acids and Derivatives
- Fmoc-Amino Acids and Derivatives
- Pseudoproline Dipeptides
- Z-Amino Acids and Derivatives
- Amino Alcohols
- Fmoc-Amino Acids Attached to Wang Resin
- Amino Acids 2-Chlorotrityl Resin
- Other Resin and Derivatives

- Detail
Categories
- FC-G7003
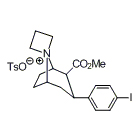
- [18F]Alzavue precursor